Research
Our most recent studies focus on the role of biological sex in immune modulation of
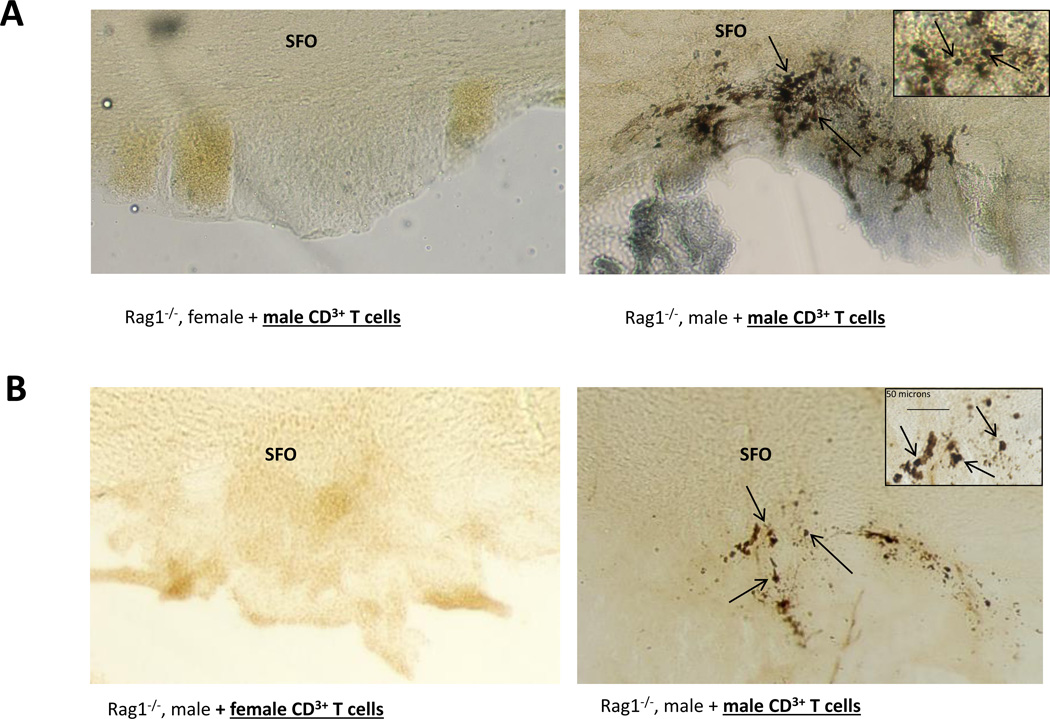
hypertension. We have found that male T cells exacerbate angiotensin II-induced
hypertension in the male mouse but are unable to drive hypertension in the female
(Pollow et al., Hypertension, 2014).
Furthermore, female T cells are not able to drive the hypertension in the male mouse
(Ji et al., Hypertension, 2014;
Sandberg et al., Cell Immunology, 2015).
These findings indicate biological sex is a critical determinant in T cell differentiation
and/or function and emphasize the importance of the recent
NIH initiative
from the NIH Office of Research on Women’s Health
to balance the sex of cells and animals in preclinical studies
(Sandberg, et al., FASEB J, 2015).
Our research on 17β-estradiol regulation of AT1R function led to studies
investigating why females are protected from hypertension and chronic kidney disease.
We found ovariectomy augments the magnitude of hypertension in aging salt-sensitive rats
(Hinojosa-Laborde et al., Hypertension, 2004)
as well as the progression of renal injury in a model of renal-induced hypertension
(Ji et al., AJP: Heart, 2005).
In addition, we found 17β-estradiol protected against the progression of hypertension and
renal disease in part by down-regulating the expression of AT1Rs
(Hinojosa-Laborde et al., Hypertension, 2004
; Ji et al., AJP: Renal, 2005).
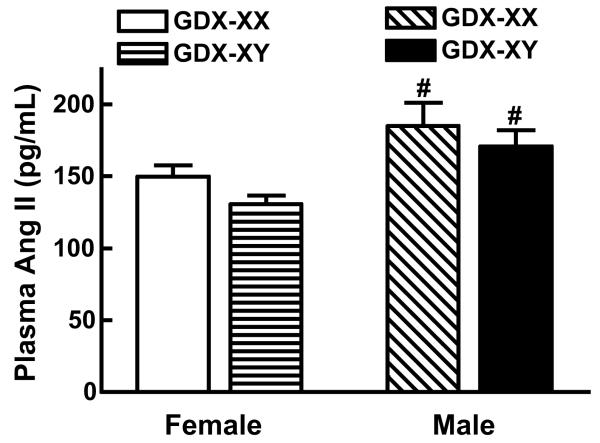
Using the unique four core genotype mouse animal model in which gonadal sex is separated from the sex
chromosomes, we found the sex chromosome complement impacts the magnitude of the
hypertension independently of gonadal hormones
(Ji et al., Hypertension 2010),
which was the report of a sex chromosome effect in hypertension.
During our laboratory's research on 17β-estradiol regulation of angiotensin type 1
receptors (AT1Rs) in the adrenal, we discovered that RNA binding proteins regulate
receptor function by selectively binding within exon 2 of the AT1aR 5'-leader sequence
(Wu et al., Endocrinology, 2003)
and that this translational regulation is mediated by a short open reading frame (sORF) in exon 2
(Ji et al, JBC 2004).
Furthermore, we found this new mechanism of posttranscriptional regulation is shared
with other peptide hormone G protein-coupled receptors
(Wu et al., Neuroendocrinology, 2004).
More recently, this research direction has led to the exciting
discovery that a seven amino acid peptide (PEP7) encoded with a short open reading
frame within exon 2 is a selective inhibitor of AT1aR signaling; PEP7 inhibits the Erk1/2
but not the classic inositol trisphosphate pathway
(Liu et al., AJP: Reg, 2014).
We have recently found that the PEP7 sORF facilitates angiotensin II-induced AT1R
internalization. These findings suggest that we have uncovered a new mechanism
governing agonist-induced AT1aR cellular trafficking that could have implications not only
for regulation of AT1aR signaling cascades but also for other trafficking proteins that
contain an upstream sORF within their 5'LS.
Ovarian hormone loss is associated with higher blood pressure. Prospective
population studies show that postmenopausal women have higher arterial
pressure than age-matched premenopausal women. Women with premature
ovarian failure also have a higher incidence of hypertension than age-matched
premenopausal women. Blood pressure is also increased in numerous animal
models of ovarian hormone loss, and estrogen treatment consistently reverses
this effect. Women who have undergone bilateral salpingo-oophorectomy prior to
menopause are at increased risk of developing cognitive impairment and
dementia than age-matched women, with greater risk the younger the age at
oophorectomy. Hypertension is also a risk factor for cognitive impairment. The
incidence of dementia is higher in individuals with hypertension, and decrements
in learning and memory have been observed in hypertensive patients
independently of demographic and psychosocial factors. We are studying the
association of blood pressure, cognition and physical activity and the role of the
renin angiotensin system in modulating learning and memory as well as the
cardiovascular and neuronal protective effects of aerobic exercise in human
subject studies as well as in animal models.
